The $1.8 Billion AI Doctor Fraud: Decoding the Propaganda Playbook Behind MEDVi
Russell Brunson’s breakdown of the MEDVi case is required watching for any marketer using AI-generated content, testimonials, or third-party authority in their campaigns. By the end, you’ll understand how manufactured expert credibility operates at industrial scale, why the regulatory machinery is already in motion, and exactly where the compliance lines fall. The lesson isn’t just about fraud — it’s the oldest influence technique in marketing, running at AI speed.
- Start with the surface narrative the New York Times published on April 2nd: Matthew Gallagher built a $1.8 billion telehealth company called MEDVi with two employees and $20,000, using AI tools — ChatGPT, Claude, Grok, MidJourney, Runway — to sell compounded GLP-1 weight loss drugs online, cheaper than Ozempic and Wegovy. The Times verified the financials. LinkedIn declared it the AI success story of the decade.

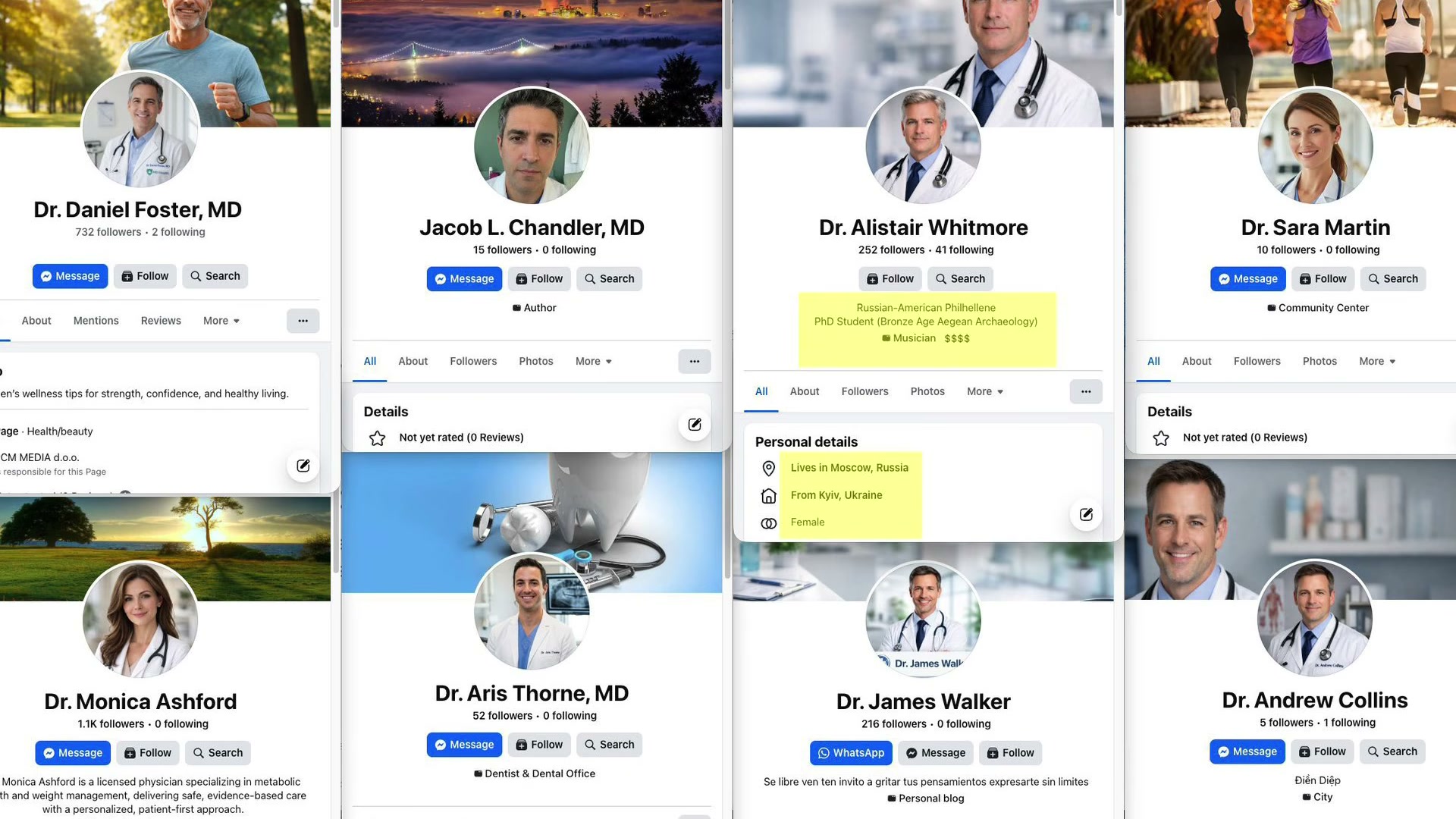
- Dig beneath the headline to find the actual acquisition engine. Gallagher and his affiliates created over 800 Facebook profiles impersonating doctors — fabricated identities built on AI-generated headshots, professional bios, and medical credentials that didn’t exist. These profiles ran thousands of ads promoting MEDVi’s weight loss drugs to real consumers who believed they were receiving physician recommendations.

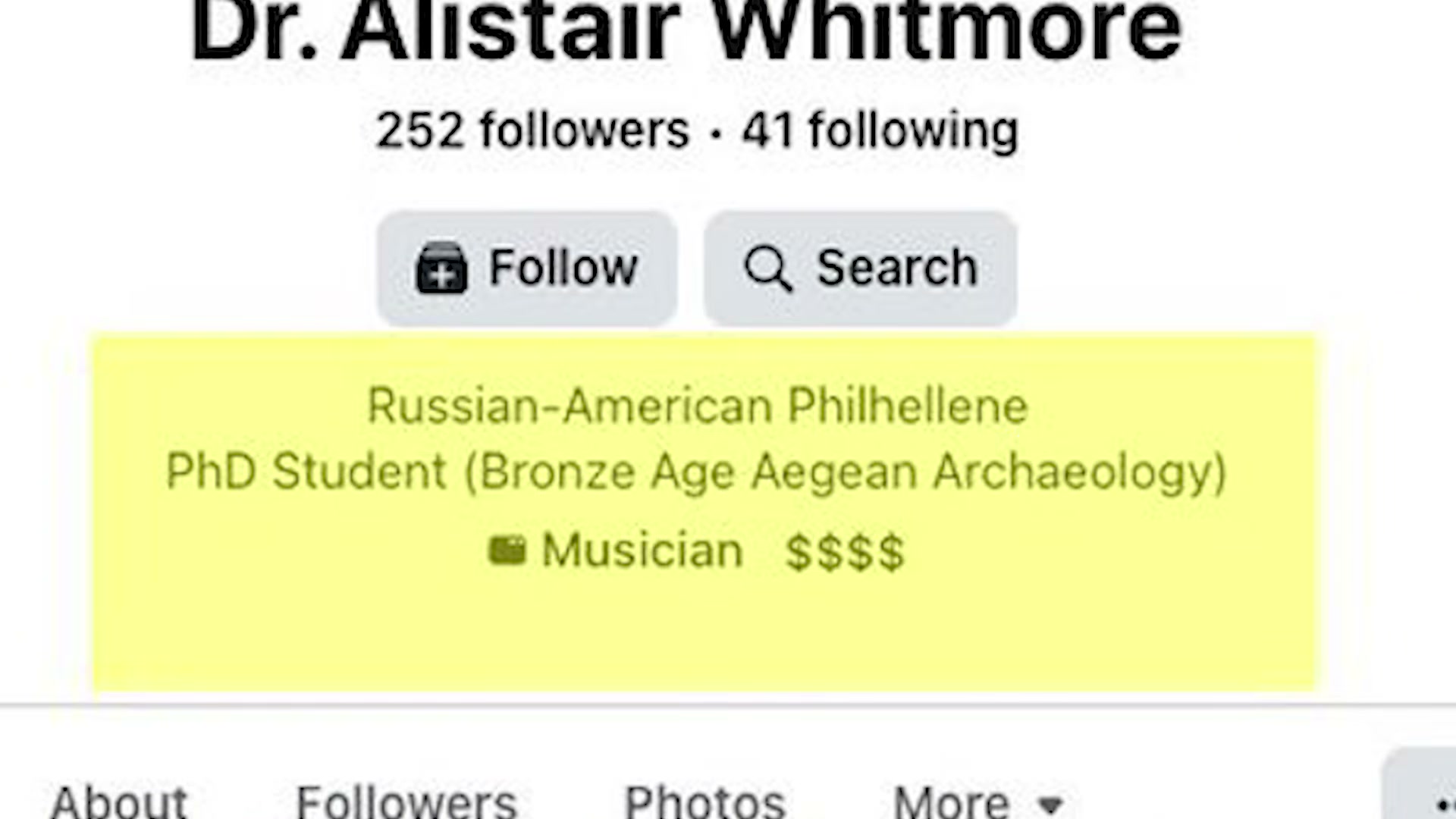
- Examine the MEDVi website’s doctor roster: professional profile photos and credentials attached to people who don’t exist. When researchers cross-referenced the profiles, “Dr. Alistair Whitmore” — supposedly a weight loss physician — turned out to be a Facebook account listing its owner as a Russian-American PhD student specializing in Bronze Age Aegean archaeology. Another fake doctor’s contact details resolved to a clothing store in the Republic of Congo.
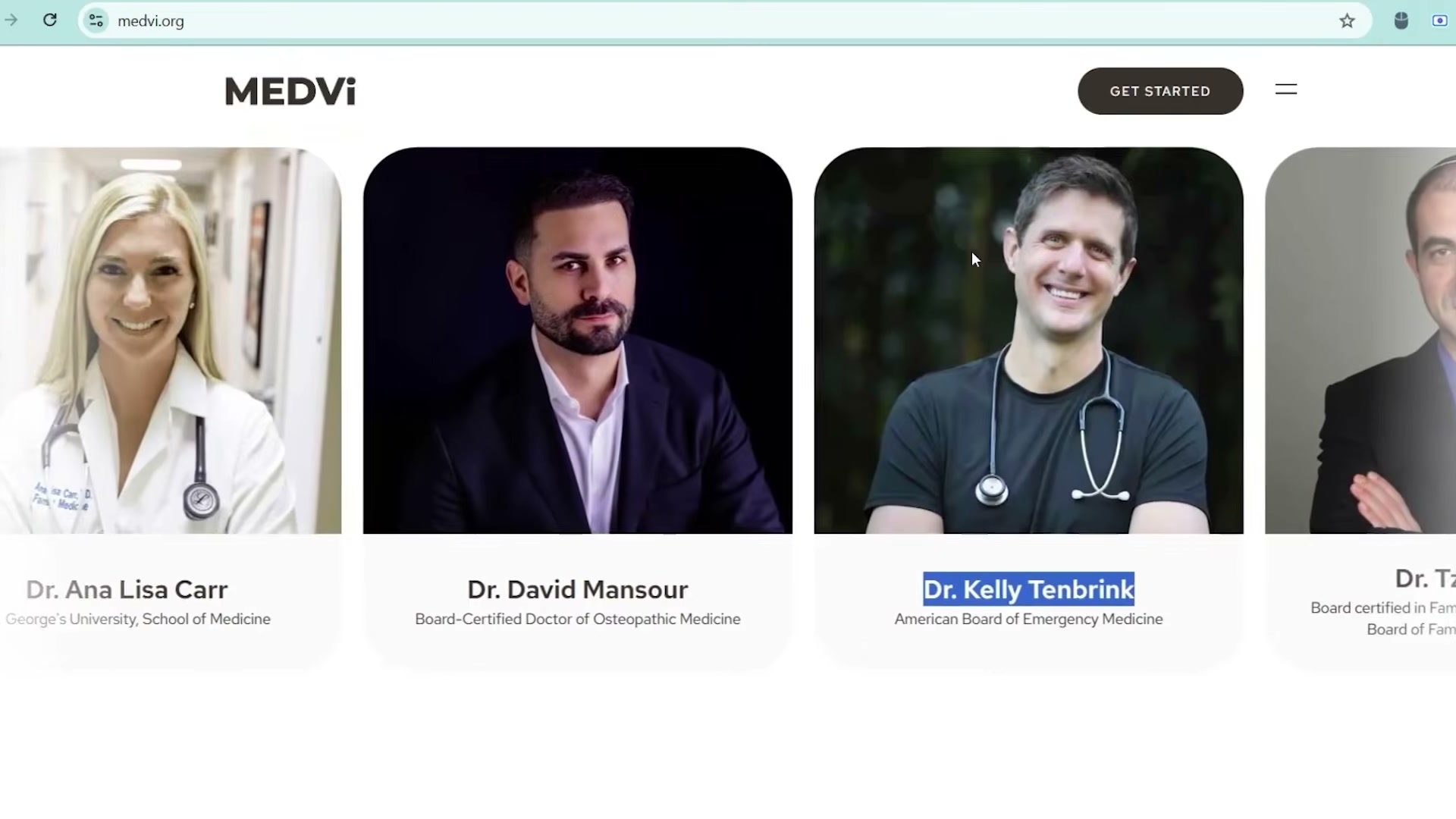
- At peak operation, 5,000 active ad campaigns on Facebook linked to MEDVi, all running under fake doctor personas, all targeting real patients. Sustaining 800+ synthetic identities at that volume required AI — no human affiliate operation could generate and manage that surface area manually.
-
Trace the FDA’s response, which came six weeks before the Times profile. On February 20th, 2026, FDA Warning Letter 721455 flagged MEDVi for falsely implying it compounded its own drugs, and for language like “same active ingredient as Ozempic” — phrasing that implies FDA approval for a product that has none. The FDA noted explicitly that the cited violations were not exhaustive.
-
Factor in the patient exposure. MEDVi’s clinical partner Openloop suffered a data breach in January 2026, with a hacker claiming records from 1.6 million patients — names, addresses, dates of birth, medical histories, treatment history. Real people who trusted fake doctors, viewed fabricated testimonials, and took drugs the FDA had already flagged.

-
Connect the technique to Edward Bernays, who codified manufactured third-party authority in 1920: people don’t trust companies or ads, but they trust experts. Bernays engaged real physicians to anchor product recommendations — doctors endorsing heavier breakfasts conveniently tied to bacon and eggs, and cigarettes promoted for weight management. The persuasion architecture MEDVi deployed is structurally identical.
-
Note the single variable separating Bernays from Gallagher: Bernays used real doctors. Gallagher fabricated them with AI. The underlying influence mechanism is the same. The legal standing is not.
- Register Brunson’s projection: FTC shutdown within 12 months, grounded in a National Consumers League filing requesting formal FTC investigation. The compounding regulatory exposure — FDA warning letter, data breach, documented fake-doctor ad network, fabricated before-and-after testimonials — spans multiple enforcement bodies simultaneously.
Warning: this step may differ from current official documentation — see the verified version below.
- Apply the compliance baseline Brunson states directly: real testimonials with documented results, only substantiated claims, and FTC endorsement guidelines treated as hard constraints rather than soft guidance. The AI tools that built MEDVi’s scale are widely available. So are the enforcement consequences for deploying them without authorization.
How does this compare to the official docs?
The FTC’s endorsement and testimonial guidelines, the FDA’s compounded drug marketing rules, and the full text of Warning Letter 721455 fill in the legal specifics the video surfaces but doesn’t map in granular detail — and Act 2 runs each one directly against what MEDVi did.
Here’s What the Official Docs Show
Act 1 mapped the MEDVi case as Russell Brunson presented it — compellingly and with the right regulatory instincts. This act layers in the primary source record: the actual FDA warning letter, FTC enforcement frameworks, and compounding drug marketing rules that define exactly where the legal exposure lives.
Step 1 — The surface narrative (NYT, April 2, 2026)
No official documentation was found for this step —
proceed using the video’s approach and verify independently.
Step 2 — 800+ fake Facebook doctor profiles driving acquisitions
No official documentation was found for this step —
proceed using the video’s approach and verify independently.
Step 3 — MEDVi’s fabricated doctor roster
No official documentation was found for this step —
proceed using the video’s approach and verify independently.
Step 4 — 5,000 active ad campaigns under synthetic identities
No official documentation was found for this step —
proceed using the video’s approach and verify independently.
Step 5 — FDA Warning Letter 721455 (February 20, 2026)
No official documentation was found for this step —
proceed using the video’s approach and verify independently.
As of April 14, 2026, FDA warning letter records are publicly searchable via the FDA’s Warning Letters database. The video’s citation of Warning Letter 721455 is specific and verifiable — readers should pull the primary document directly rather than relying on any secondary account, including this one.
Step 6 — Openloop data breach, 1.6 million patient records
No official documentation was found for this step —
proceed using the video’s approach and verify independently.
Step 7 — Edward Bernays and manufactured third-party authority
No official documentation was found for this step —
proceed using the video’s approach and verify independently.
Step 8 — Real doctors vs. AI-fabricated doctors: same architecture, different legal standing
No official documentation was found for this step —
proceed using the video’s approach and verify independently.
Step 9 — FTC shutdown projection, National Consumers League filing
No official documentation was found for this step —
proceed using the video’s approach and verify independently.
Step 10 — Compliance baseline: real testimonials, substantiated claims, FTC endorsement guidelines
No official documentation was found for this step —
proceed using the video’s approach and verify independently.
The FTC’s Guides Concerning the Use of Endorsements and Testimonials in Advertising (16 CFR Part 255) is the governing document for every testimonial-based health marketing claim in the U.S. It is publicly available and should be read in full before any AI-assisted testimonial campaign goes live.
Useful Links
- FDA Warning Letters Database — Searchable index of all FDA warning letters, including those covering compounded drug marketing violations
- FTC Endorsement Guides: 16 CFR Part 255 — The binding federal standard for testimonial and endorsement disclosures in advertising
- FDA — Compounded Drug Products FAQ — FDA’s official guidance on lawful compounding and prohibited marketing claims, including comparative drug language
- FTC Health Claims Guidance — Plain-language FTC explainer on what constitutes a compliant health-related endorsement or testimonial

0 Comments